This article is an installment of Future Explored, a weekly guide to world-changing technology. You can get stories like this one straight to your inbox every Thursday morning by subscribing here.
A newly trialed brain implant injects tiny doses of up to 20 different drugs into brain tumors to compare how well they work. Tech like this could help doctors rapidly screen lots of treatments to find the drug most likely to help — and give patients their best shot right away.
Gliomas 101
About 20,000 people are diagnosed with gliomas in the US every year, making them one of the most common types of brain tumor. They’re also one of the deadliest — more than half of gliomas are an aggressive type called “glioblastoma.” Just 5% of people with this type survive for five or more years after diagnosis.
Gliomas are often treated with surgery to remove as much of the tumor as possible, followed by another therapy to kill any cells that might have been missed. It’s hard for doctors to know right away which therapy to administer, though, and given the aggressiveness of many gliomas, they might not have time to try alternatives.
“In order to make the greatest impact on how we treat these tumors, we need to be able to understand, early on, which drug works best for any given patient,” said Pierpaolo Peruzzi, a neurosurgeon at Brigham and Women’s Hospital. “The problem is that the tools that are currently available to answer this question are just not good enough.”
The microdevice
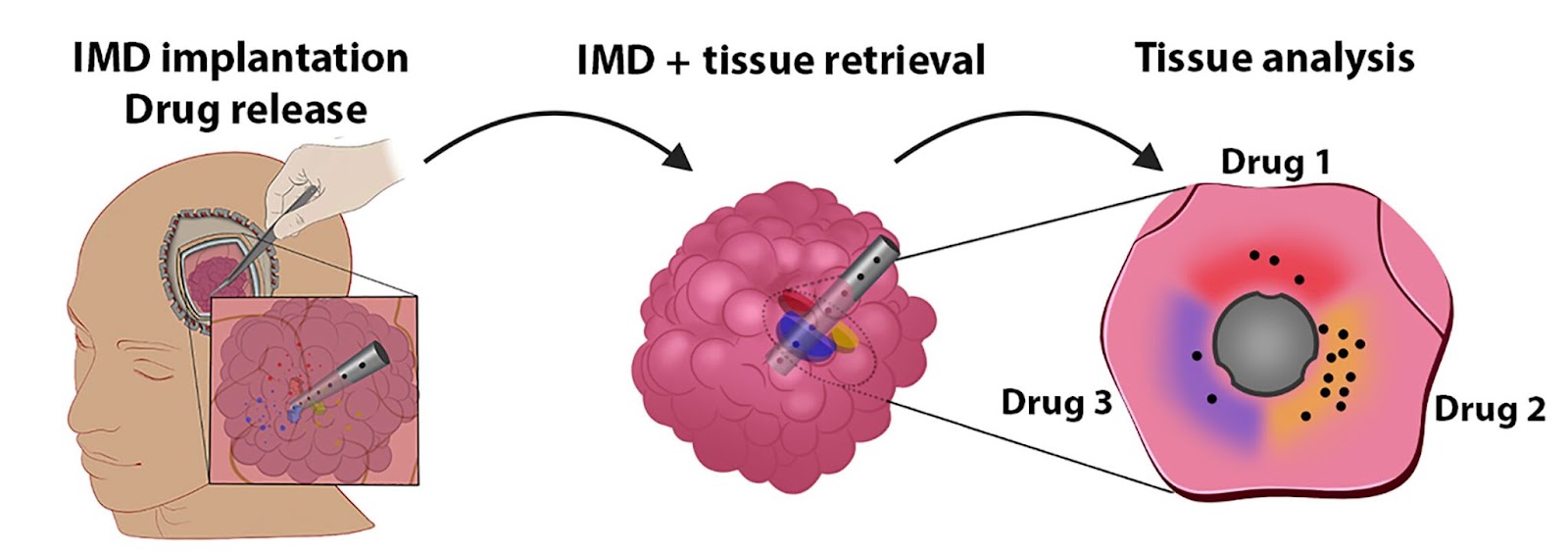
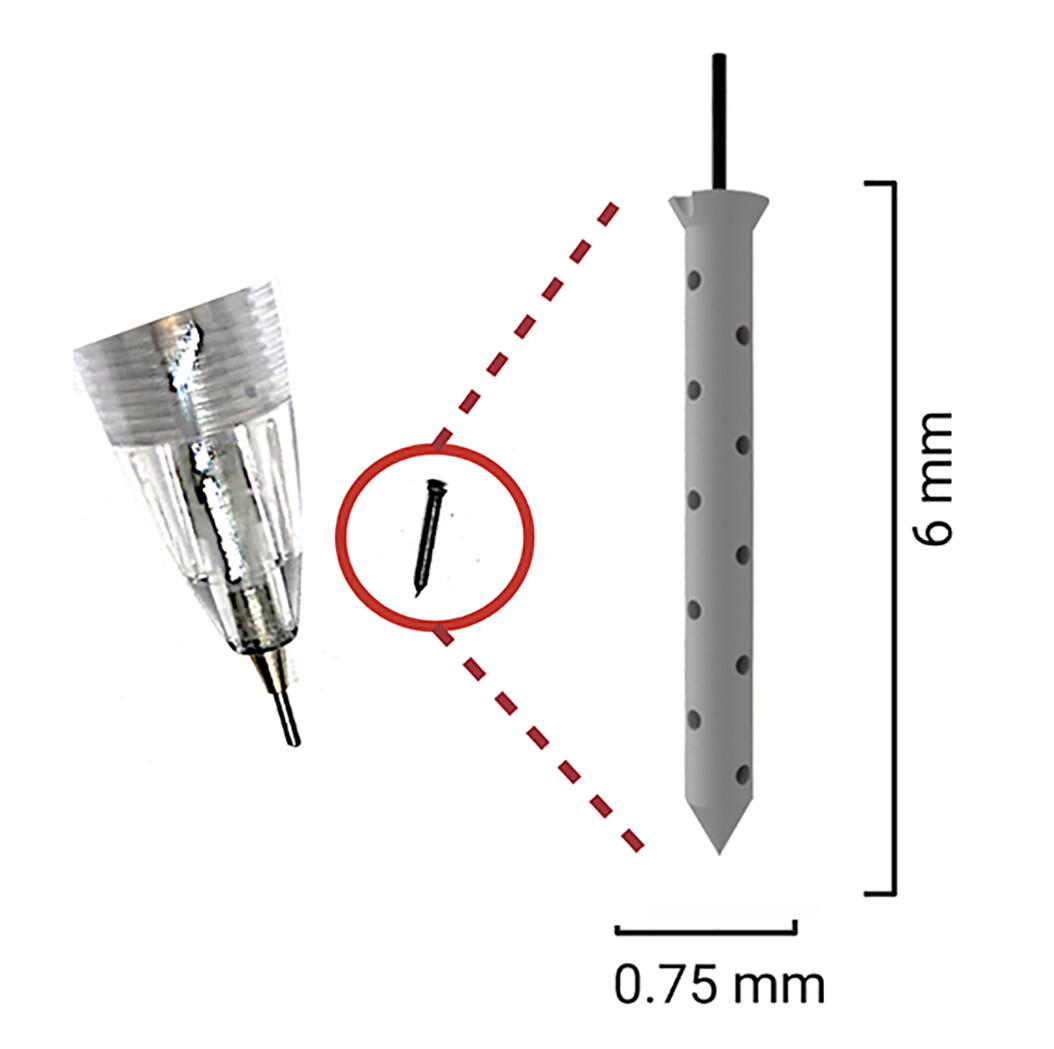
Peruzzi and his colleagues have now developed a tiny implant that can be stuck inside a tumor — called an “intratumoral microdevice” (IMD) — during surgery. The implant is only the size of a grain of rice, but it can administer up to 20 drugs to different parts of the tumor over the course of the operation.
The implant is then removed, along with the tumor tissue, which is frozen and sent to a lab for analysis. Researchers can then look for signs of cell death and DNA damage in each part of the tissue to determine which drugs are most likely to work against that patient’s cancer.
“This is not in the lab, and not in a petri dish,” said Peruzzi. “It’s actually in real patients in real time, which gives us a whole new perspective on how these tumors respond to treatment.”
In a phase 1 trial, the researchers tested their microdevice on six patients undergoing glioma removal surgery, and it didn’t cause any unwanted side effects.
The trial also demonstrated that integrating the microdevice into the standard surgery wouldn’t be a major burden on doctors and wouldn’t significantly increase costs for patients — though it would vary depending on the hospital, they estimated an increase of $7,800 at Brigham and Women’s Hospital, which equates to 7.5% of the total surgery cost.
While data from this trial wasn’t taken into consideration when deciding how to treat the patients, two later received one of the meds, called “TMZ,” that had been loaded into the microdevice.
One of those patients didn’t respond to the treatment — and it turns out they also showed poor response to the drug in the trial’s tissue analysis. The other patient responded well to TMZ, and they showed a marked response to the drug during the trial.
A third patient received the drug prior to undergoing the surgery. Their doctor already knew TMZ wasn’t working, but the tissue analysis also suggested that it wouldn’t be effective for that patient.
The example of a single drug tested on three patients is, of course, far too little data to draw any conclusions about how well this device might ultimately improve treatment, and larger trials with prespecified study plans will be needed.
Looking ahead
If a device like this is able to improve patient outcomes, it’s not hard to imagine it being used to test treatments for other types of brain tumors or during drug development, potentially helping identify cancer patients most likely to benefit from experimental therapies.
For now, the researchers are exploring whether it’d be worthwhile to implant their microdevice into gliomas a few days prior to the removal surgery. This would give it more time to act on the tumor tissue, but would also increase costs and require patients to undergo a second, albeit less invasive, operation.
Ultimately, though, they’re encouraged by the phase 1 trial results and what their device could mean for patients diagnosed with gliomas in the future.
“We’re optimistic that this is a new generation approach for personalized medicine,” said Peruzzi. “The ability to bring the lab right to the patient unlocks so much potential in terms of the type of information we can gather, which is new and exciting territory for a disease that has very few options at present.”
We’d love to hear from you! If you have a comment about this article or if you have a tip for a future Freethink story, please email us at [email protected].